"Importantly, physicians can continue to use the Proclaim and Infinity systems. Boston Scientific will pay Nevro 85 million and will receive a license to practice paresthesia-free therapy at frequencies below 1,500Hz, according to an Aug. Spinal cord stimulator procedures are largely done in private hospitals where they can cost more than 50,000 per procedure. "No products need to be returned to the company or replaced," according to Abbott. Abbotts Proclaim and Infinity devices, which are used in spinal cord and deep brain stimulation, have a technical malfunction that causes them to be temporarily turned off when patients have MRI scans. The litigation concerns high-frequency spinal cord stimulation technology for pain relief using 10,000Hz spinal cord stimulator technology. Patients should also ensure their PC devices have been updated to the latest version in their smartphone’s app store.Ībbott emphasized in an emailed statement that this Class I recall did not require all impacted devices to be sent back to the manufacturer. Paired Bluetooth connections between IPGs and PC devices should not be deleted, for example, and PC devices should not be altered in any way while the IPG is in MRI mode. The letter, available here in full, includes recommendations for physicians to reduce the risk of IPG replacement surgery. Specific product names include the Proclaim XR 5 IPG, Proclaim XR 7 IPG, Proclaim Plus 5 IPG, Proclaim Plus 7 IPG, Proclaim DRG IPG, Infinity 5 IPG and Infinity 7 IPG.Ībbott previously alerted customers about this issue back in July with an Urgent Medical Device Correction. The IPGs were distributed to customers from November 2015 to June 2023. Spinal cord stimulation, which was first introduced in 1967, is known to be an effective and minimally invasive method of treating intractable diseases, such as the complex regional pain syndrome and the post-spine-surgery syndrome. 23, 2022 /PRNewswire/ - Abbott (NYSE: ABT) today announced that the U.S. 
Spinal cord stimulation may be: An effective alternative when conventional therapies and systemic pain medications provide. Has anyone had a abbot spinal cord stimulator put in Posted by jnd2023 jnd2023, Feb 13 1:41pm. Instead of pain, patients may feel pain relief. Abbott spinal cord stimulation FDA approval neurostimulation. 
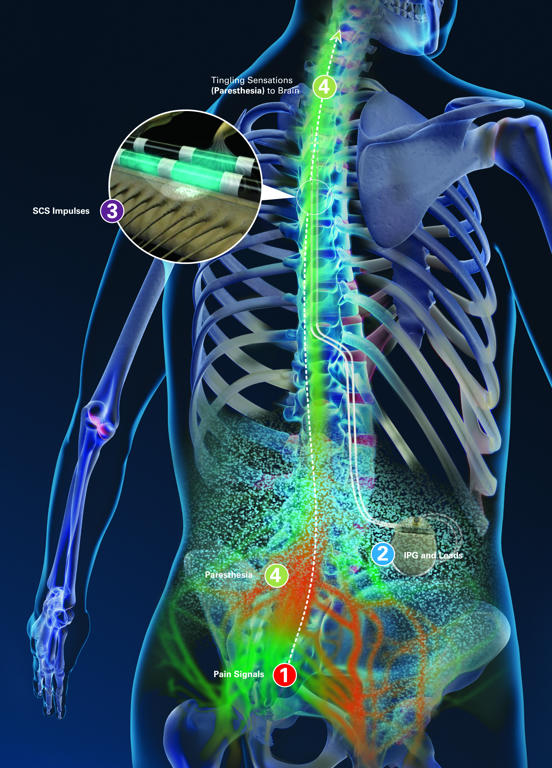
Pain signals are inhibited before they reach the brain. Abbotts spinal cord stimulators score FDA approval to treat nonsurgical back pain. Abbotts low-energy spinal cord stimulation is a non-opioid solution offering an innovative approach to chronic pain management. A total of 186 incidents and 73 patient injuries have been reported due to these issues. An implantable spinal cord stimulator delivers small electrical signals through a lead implanted in the epidural space.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
